|
4/20/2023 0 Comments Ir spectrum functional groups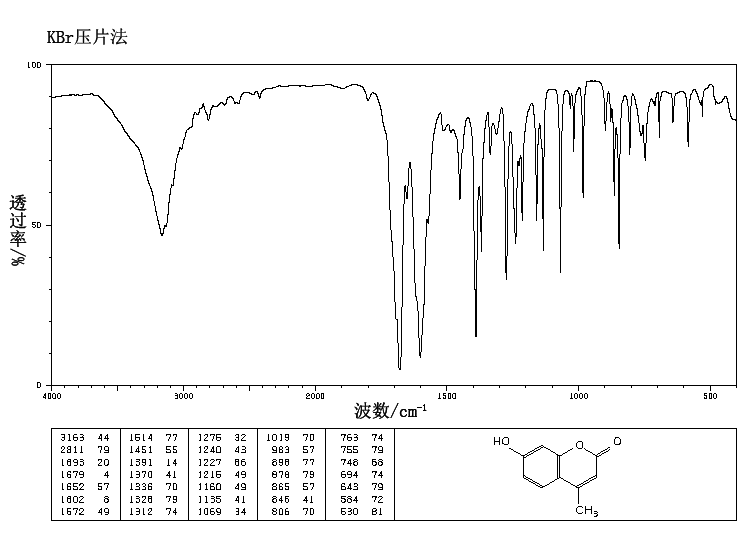 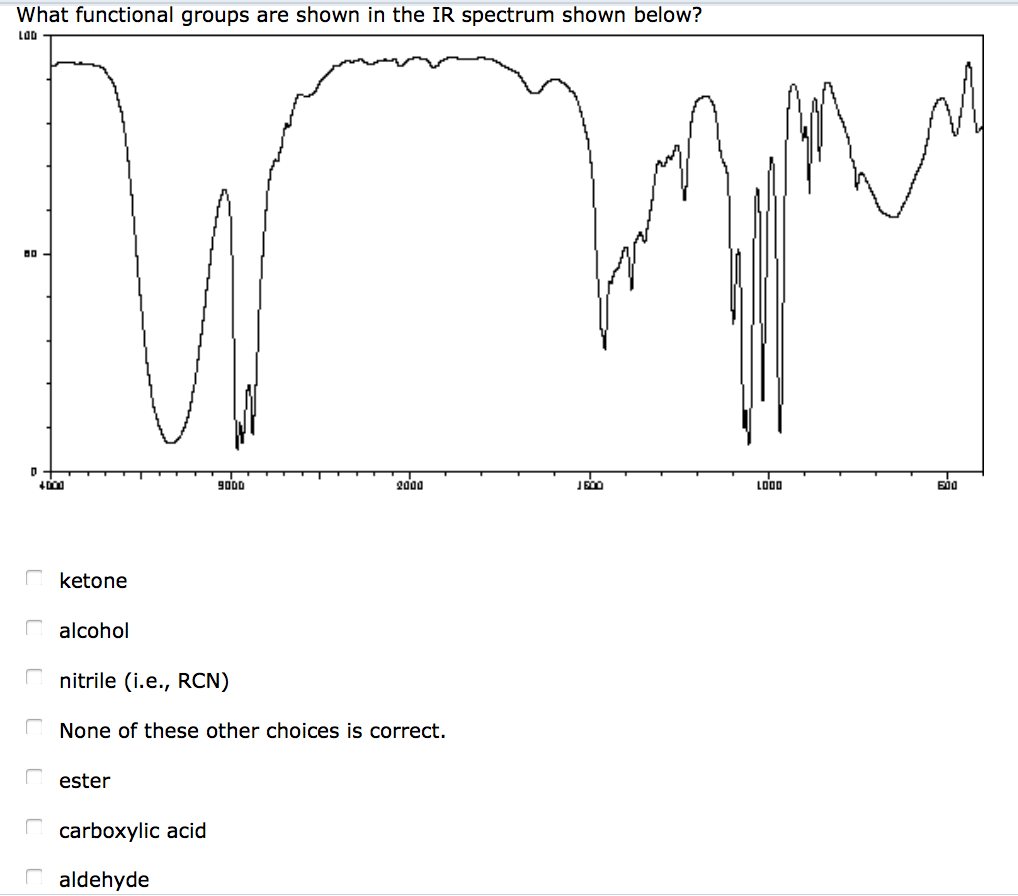
Ī molecule can vibrate in many ways, and each way is called a vibrational mode. A permanent dipole is not necessary, as the rule requires only a change in dipole moment. In order for a vibrational mode in a sample to be "IR active", it must be associated with changes in the dipole moment. Thus, the frequency of the vibrations are associated with a particular normal mode of motion and a particular bond type. The resonant frequencies are also related to the strength of the bond and the mass of the atoms at either end of it. when the molecular Hamiltonian corresponding to the electronic ground state can be approximated by a harmonic oscillator in the neighborhood of the equilibrium molecular geometry, the resonant frequencies are associated with the normal modes of vibration corresponding to the molecular electronic ground state potential energy surface. In particular, in the Born–Oppenheimer and harmonic approximations, i.e. The names and classifications of these subregions are conventions, and are only loosely based on the relative molecular or electromagnetic properties.ģD animation of the symmetric stretch-compress mode of the C–H bonds of bromomethane The region from 2–130 cm −1, bordering the microwave region, is considered the terahertz region and may probe intermolecular vibrations. The far-infrared, approximately 400–10 cm −1 (25–1,000 μm) has low energy and may be used for rotational spectroscopy and low frequency vibrations. The mid-infrared, approximately 4,000–400 cm −1 (2.5–25 μm) is generally used to study the fundamental vibrations and associated rotational–vibrational structure. The higher-energy near-IR, approximately 14,000–4,000 cm −1 (0.7–2.5 μm wavelength) can excite overtone or combination modes of molecular vibrations. The infrared portion of the electromagnetic spectrum is usually divided into three regions the near-, mid- and far- infrared, named for their relation to the visible spectrum. Two-dimensional IR is also possible as discussed below. A common laboratory instrument that uses this technique is a Fourier transform infrared (FTIR) spectrometer. Units of IR wavelength are commonly given in micrometers (formerly called "microns"), symbol μm, which are related to the wavenumber in a reciprocal way. Typical units of wavenumber used in IR spectra are reciprocal centimeters, with the symbol cm −1. frequency, wavenumber or wavelength on the horizontal axis. 
An IR spectrum can be visualized in a graph of infrared light absorbance (or transmittance) on the vertical axis vs. The method or technique of infrared spectroscopy is conducted with an instrument called an infrared spectrometer (or spectrophotometer) which produces an infrared spectrum. It can be used to characterize new materials or identify and verify known and unknown samples. It is used to study and identify chemical substances or functional groups in solid, liquid, or gaseous forms. Infrared spectroscopy ( IR spectroscopy or vibrational spectroscopy) is the measurement of the interaction of infrared radiation with matter by absorption, emission, or reflection. Here’s a video that gives a simple explanation of infrared spectroscopy.OVIRS instrument of the OSIRIS-REx probe is a visible and infrared spectrometer The C=O and C-O absorptions tell you that this is an ester. Notice how you can identify the important vibrations in the spectrum of ethyl acetate. Here’s a short table of common absorption frequencies. This makes infrared spectroscopy useful to identify functional groups in a molecule. Within a narrow range, each type of bond vibrates at a characteristic wavenumber. The spectrometer detects this absorption and records it as a peak in a plot of transmission versus frequency. The amplitude of the vibration will increase. If the frequency of the radiation matches the vibrational frequency, the bond will absorb the radiation. The alternating electrical field of the infrared radiation can interact with the alternating dipole moment. Rather, bond lengths and angles represent the average positions about which atoms vibrate.Įach vibration may cause a change in the dipole moment of the molecule. Molecules do not have rigid bond lengths and bond angles. 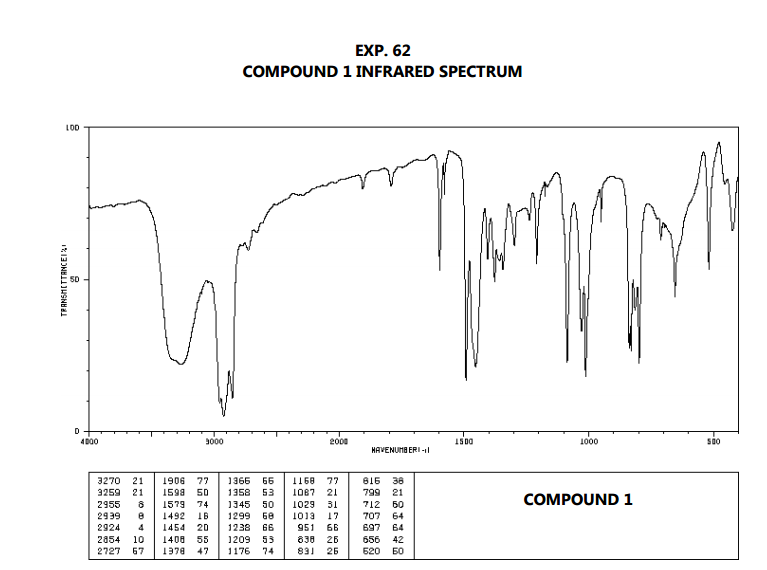
So the wavenumbers range from 500 to 4000 cm⁻¹. #"wavenumber" = "frequency"/"speed of light" = ν/c# These frequencies are expressed as wavenumbers: In organic chemistry, this corresponds to frequencies of 15 to 120 THz. Vibrating bonds absorb infrared energy at a frequency that corresponds to the vibrational frequency of the bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
